 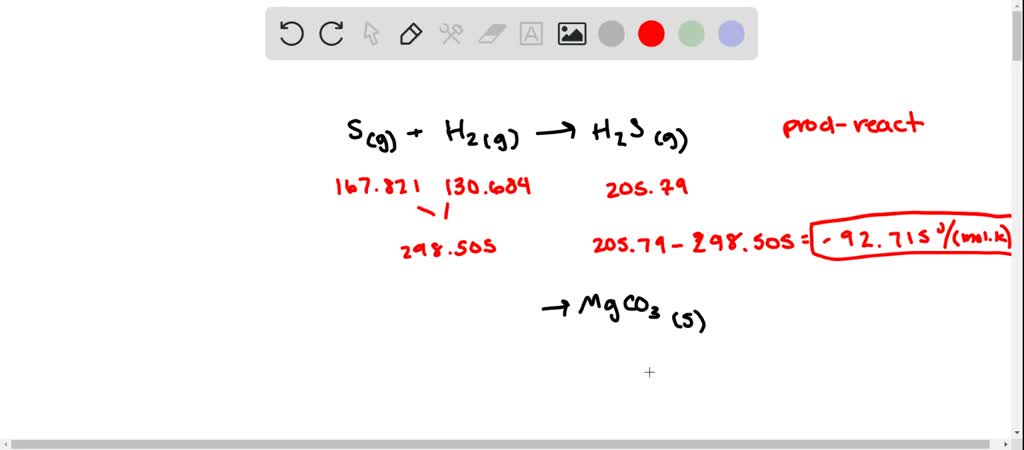
Given Kf for water = 1.86 KKg mor Login Molar mass of MgBr2 is 184.1130 g/mol Compound name is magnesium bromide Get control of 2022! Track your food intake, exercise, sleep and meditation for free. Given Kf for water = 1.86 KKg mor Q6 Calculate the freezing point of an aqueous solution containing 10.50 g of MaBrz in 200 gof water (molar mass of MgBr2 184 g mol 1 ). Is the Reaction Exothermic or Endothermic? MgCl 2 (s) 1 mol …Q6 Calculate the freezing point of an aqueous solution containing 10.50 g of MaBrz in 200 gof water (molar mass of MgBr2 184 g mol 1 ). Thermodynamics of the reaction can be calculated using a lookup table. Salt Nacl Table Salt Common Salt Broncho Saline NaCl Molar Mass NaCl Bond Polarity NaCl Oxidation Number. 
Is the Reaction Exothermic or Endothermic? MgO (s) 1 mol-601.7 …Magnesium bromide | MgBr2 or Br2Mg | CID 82241 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological. Hoh Aqua H₂O Oxidane Pure Water Hydroxic Acid Hydrogen Oxide H2O Molar Mass H2O Oxidation Number. 
Thermodynamics of the reaction can be calculated using a …MgBr2 Molar Mass MgBr2 Oxidation Number Magnesium Chloride - MgCl 2 Mgcl2 Magnesium Chloride Anhydrous MgCl2 Molar Mass MgCl2 Oxidation Number Reaction Expressions Equilibrium Constant & Reaction Quotient Rate of Reaction Calculate Reaction Stoichiometry Calculate Limiting ReagentMgBr2 Molar Mass MgBr2 Oxidation Number. Cabr2 CaBr2 Molar Mass CaBr2 Oxidation Number. Mots clés : alcools en solutions aqueuses, entropies molaires partielles en excès, entropie d'interaction, schémas de mélange, valeurs relatives d'hydrophobicité.Science Chemistry Chemistry questions and answers In the following reaction, 35.0 mL of 0.150 M magnesium bromide solution reacts with excess silver nitrate to form a precipitate. De plus, sur la base des valeurs de S E AL≪L et de H E AL≪L, on a établi que l'ordre de l'hydrophobicité relative des alcools est méthanol < éthanol < propan-2-ol < propan-1-ol. Les conclusions auxquelles on était arrivé antérieurement en se basant uniquement sur des valeurs de H E AL et H E AL≪L sont confirmées. Utilisant ces quantités, on a étudié les schémas de mélange dans alcools en solutions aqueuses. La valeur de S E AL≪L définie plus haut correspond à l'effet d'une addition d'alcool sur la situation entropique réelle d'alcool existant en solution. On a par la suite calculé d'une façon numérique l'interaction entropique S E AL≪L = N( S E AL/ n AL) p,T,n W où n AL = quantité d'alcool, n W = quantité d'eau et N = quantité totale de solution. 74, 713 (1996)) dans ce laboratoire, on a calculé les entropies molaires partielles en excès des alcools, S E AL, où AL représente un alcool. Utilisant les enthalpies molaires partielles en excès de l'alcool, H E AL, déterminées antérieurement (Can. Résuméĭans le travail précédent, on a rapporté des valeurs de potentiels chimiques qui ne dépendant d'aucun modèle pour des mélanges binaires aqueux de méthanol, d'éthanol, de propan-1-ol, de propan-2-ol, de butan-1-ol et d'hexan-1-ol, à 25☌, sur l'ensemble des plages de composition. Key Words: aqueous alcohols, excess partial molar entropies, entropic interaction mixing schemes, hydrophobicity ranking. Furthermore, the order of the relative hydrophobic nature of alcohols is established from the behaviour of S E AL≪L and of H E AL≪L as methanol < ethanol < 2-propanol < 1-propanol. The earlier conclusions, which used H E AL and H E AL≪L alone, are confirmed. Using these quantities, the mixing schemes in aqueous alcohols have been studied. S E AL≪L signifies the effect of addition of AL upon the entropic situation of existing AL in solution. We then calculated, numerically, the entropic interaction, S E AL≪L = N( S E AL/ n AL) p,T,n W, where n AL is the amount of AL, n W is the amount of H 2O, and N is the total amount of solution. 74, 713 (1996)), we have calculated excess partial molar entropies for the alcohols, S E AL, where AL stands for an alcohol. Using alcohol excess partial molar enthalpies, H E AL, determined earlier in this laboratory (Can. In the preceding paper, we reported the values of model-free chemical potentials for aqueous methanol, ethanol, 1-propanol, 2-propanol, 1-butanol, and 1-hexanol at 25☌ over the entire compositional region. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
